Schistosomiasis Control Initiative
Giving What We Can no longer conducts our own research into charities and cause areas. Instead, we're relying on the work of organisations including J-PAL, GiveWell, and the Open Philanthropy Project, which are in a better position to provide more comprehensive research coverage.
These research reports represent our thinking as of late 2016, and much of the information will be relevant for making decisions about how to donate as effectively as possible. However we are not updating them and the information may therefore be out of date.
Cause Area: Parasitic Worms
We believe that the Schistosomiasis Control Initiative (SCI) is one of the most effective charities in the world. It works in a high priority area, combination deworming, which may be extremely cost-effective. SCI also has a strong track record in delivering the intervention, demonstrates a commitment to evidence and transparency, and continues to have room for more funding to be used productively.
Whether combination deworming has substantial effects on health and child development is subject to ongoing academic debate. Nevertheless, there is still strong evidence in favour of the efficacy of deworming and there is a considerable probability that the impact of deworming is quite large. As mass deworming is so inexpensive, the potential cost-effectiveness might be very high.
Deworming schoolchildren through interventions supported by SCI cost $1.26 per treatment.1 Not including the contributions of governments and other organisations, SCI spends $0.53 per treatment.2 It is difficult to compare this to estimates of cost per death prevented from bed net distributions that protect from malaria, because mortality due to worms is much lower. However, GiveWell has estimated that donations to SCI are roughly comparable to bed net distributions. Moreover, due to potential improvements in earnings later in life resulting from deworming in childhood, deworming by SCI is roughly 6 times more effective than direct cash-transfers by GiveDirectly.3
There is also some evidence suggesting that the mass drug administration (MDA) intervention carried out by SCI might be cost-effective in reducing the prevalence of malaria and HIV (see below). However, the evidence for this is not very strong.
In addition, SCI has an excellent track record of starting and assisting deworming projects in a number of countries in Sub-Saharan Africa and there appears to be a large long-term funding gap globally. For SCI as an organisation, GiveWell reports that any funds donated to SCI from now until February/March 2017 would be used for the organization’s 2017-18 budget, and that SCI would be able to productively use approximately $11.6 million in additional funds.4
What do they do?
SCI provides technical assistance to governments in Sub-Saharan Africa to implement mass drug administration (MDA) programs for schistosomiasis, soil-transmitted helminths (STHs) and other neglected tropical diseases - effectively, combination deworming programs. SCI's role consists of identifying country recipients, providing funding and advisory support, and conducting research on the programs.5
In concrete terms, SCI main operations involve the following:
- Funding governments to identify regions in need and carry out these treatment programs;
- Program management and providing technical assistance to countries, particularly to build capacity for MDA programs; and
- Conducting research and monitoring of areas treated.
SCI currently works in Burundi, Cote d'Ivoire, Democratic Republic of the Congo, Ethiopia, Liberia, Madagascar, Malawi, Mauritania, Mozambique, Niger, Nigeria, Rwanda, Senegal, Sudan, Tanzania, Uganda, Yemen, Zambia, Zanzibar6.
The MDA programs themselves are primarily carried out in areas with relatively high prevalence of schistosomiasis and/or STH infections. Albendazole is used to treat STH. Praziquantel is used to treat schistosomiasis. Combination deworming involves administering treatments for both types of parasitic worm. As the side-effects of the drugs are relatively minor, they can be administered to the whole population without the need for individual diagnosis, keeping costs low and reducing the need for skilled health professionals. MDAs can target everyone in a particular area or can specifically target at-risk groups such as children.7 Schools are often used as a distribution platform and teachers can be trained to distribute the drugs safely. As the treatment only requires drug distribution, MDA can be undertaken in relatively short timescales. The WHO recommends that MDA is carried out twice yearly in endemic areas.8
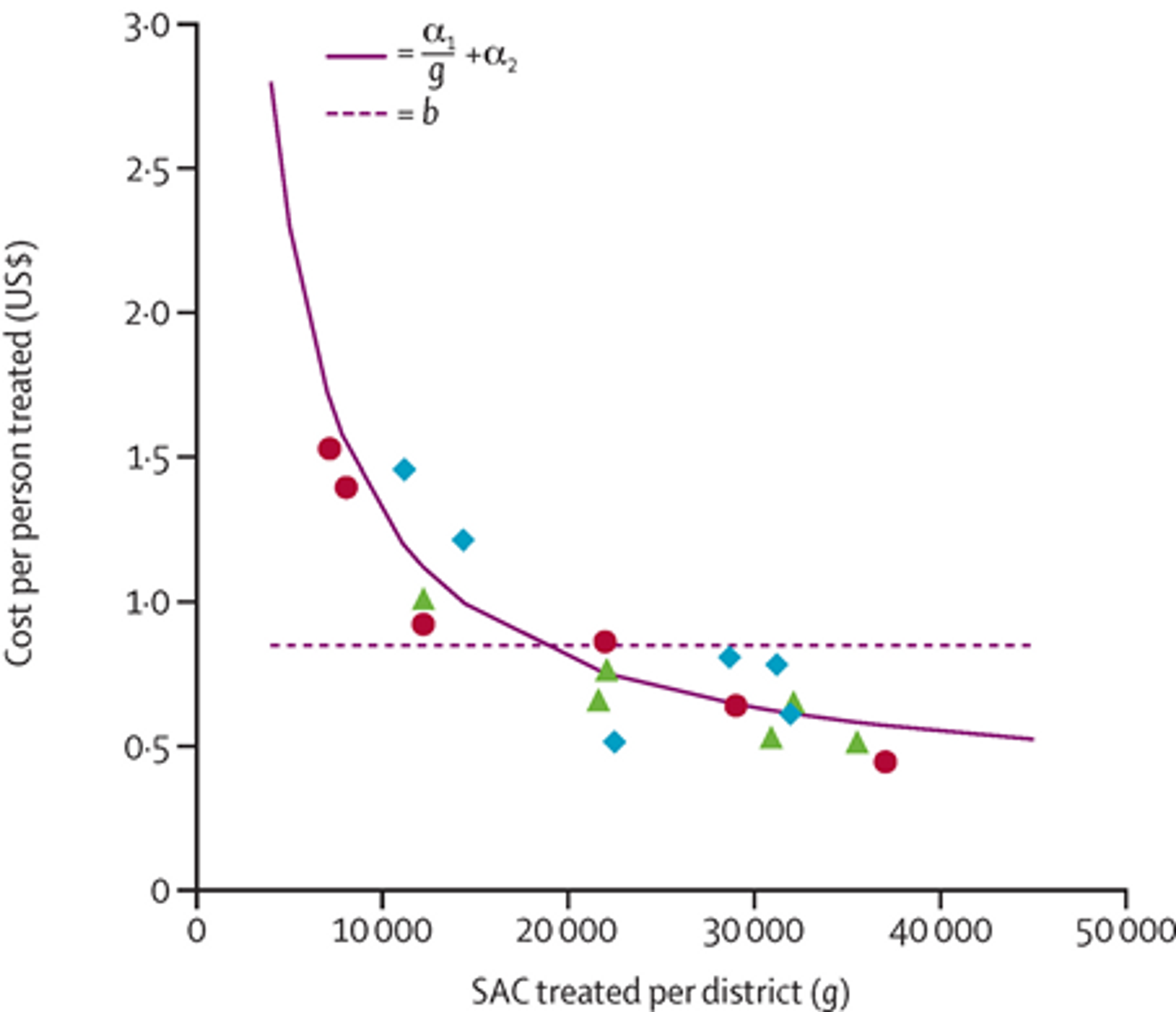
As praziquantel and albendazole (the drugs used by deworming charities) have few significant side effects,9,10 they can be safely administered to large parts of the population without the need for costly individual diagnosis that requires trained medical practitioners. GiveWell estimates that SCI can deworm a person for approximately $1.26 based on recent track records11 (including the estimated cost of teacher time, donated drugs, and local government involvement). A recent study looked at cost of treatment in Uganda for different distribution sizes and concluded that there were significant economies of scale to mass drug administration, with the average cost of STH deworming falling below $0.50 for the largest distributions (see Figure 1 below).12 It should be noted that this study was conducted by a researcher affiliated with SCI.
Figure 1: Average cost per STH treatment, by district size.13 The y-axis of figure is the cost-per-person treated of an albendazole school-based delivery programme in Uganda and the x-axis is the number of at-risk school-age children treated within each district. The negative slope shows that economies of scale are successfully being taken advantage of.
There is strong evidence that such MDA programs result in lower rates of worm infections. Two Cochrane meta-studies have concluded that deworming for schistosomiasis has a substantial impact on schistosomiasis infections, with 60% of those treated with praziquantel cured after one to two months of treatment. Moreover, the number of schistosome eggs found in the urine was reduced by 95%, helping to interrupt transmission.1415 Another meta-study, published in 2000, found that administration of albendazole was very effective in reducing levels of hookworm and ascariasis, although the median cure rate for whipworm infection was relatively low at 38%.16
Figure 2: Map of SCI’s programs 17
Cost-effectiveness
Combination deworming may be one of the most cost-effective health interventions available. Our full report on deworming illustrates our reasoning behind this conclusion. The potential impact of deworming on the long term development of children provides a strong justification for donating to a deworming charity, particularly one which is primarily focused on deworming schistosomiasis, for which the evidence is generally stronger than soil transmitted helminths.
GiveWell estimates that programs supported by SCI can deworm a person for approximately $1.26 based (including the estimated cost of SCI’s funding to country programs, SCI’s headquarters costs, cost of donated drugs, and local government involvement).18 GiveWell also estimates that SCI itself spends around $0.53 per child treated. For details of the model and assumptions used to arrive at these figures, see GiveWell’s cost-effectiveness analysis.19 SCI has recently claimed that its cost per treatment from April 2016 onwards will average $0.30 (this does not take into account other program-costs, such as the cost of drugs, teacher time and government contributions).20
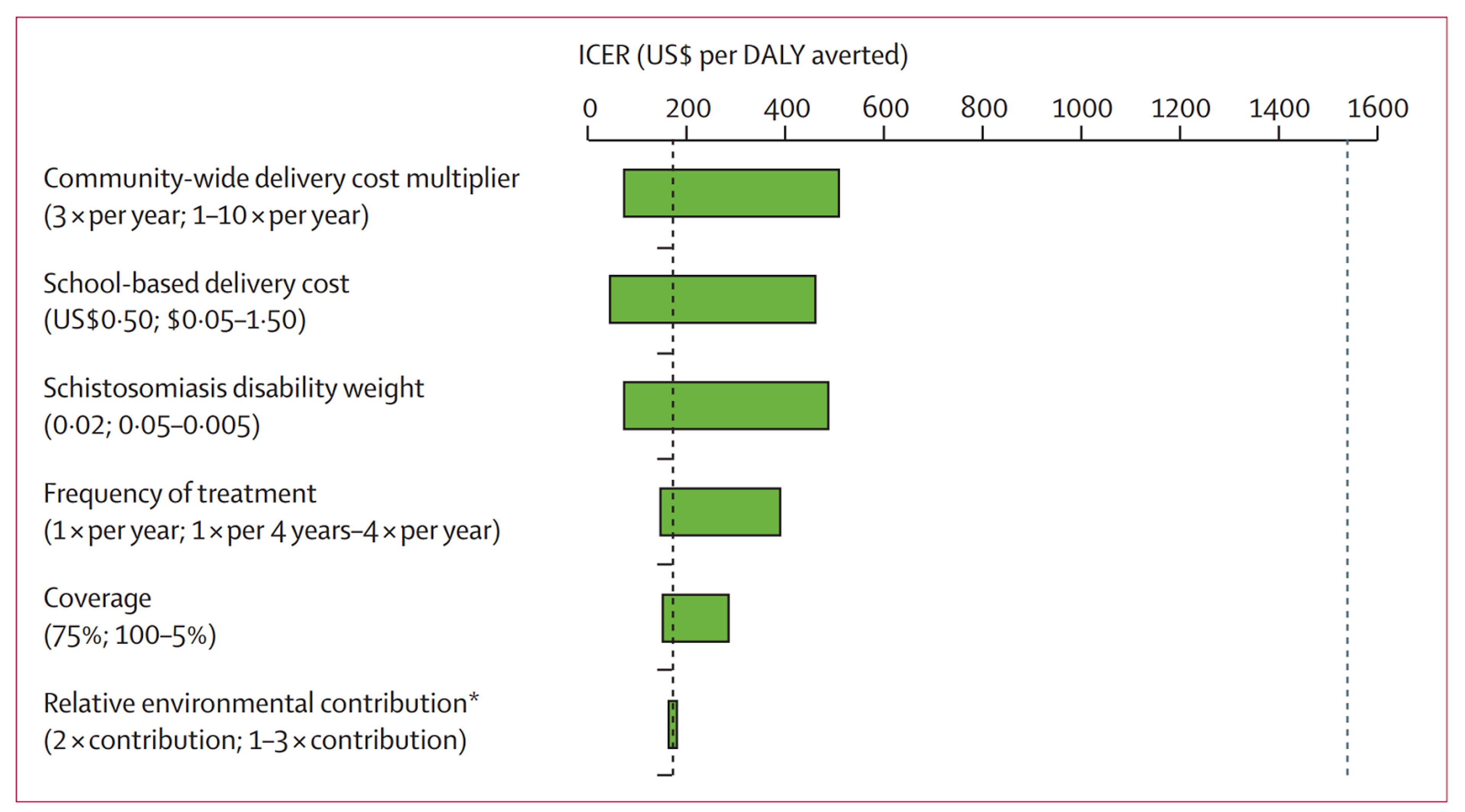
In terms of cost per life saved or cost per DALY averted, there is considerable uncertainty. A recent paper on the cost-effectiveness of school-based STH treatment programmes found that the cost per DALY averted was approximately $118, with a range of $100-500 (see Figure below).2122 However, these figures are highly uncertain, due to various challenges, including lack of data and sensitivity to many assumptions (see Figure below). It remains plausible to think, though, that the cost-effectiveness of SCI’s program is likely to be extremely high.
Figure 3 (adapted from 23): This analysis tested the effect of varying key model parameters on the incremental cost-effectiveness ratio (ICER) of expanding mass drug administration to entire communities compared with only treatment of school-aged children. The horizontal bar represents the range of ICER values for the specified range of the tested parameter. All strategies left of the dashed vertical line at US$1521 per disability-adjusted life-year (DALY) averted (2013 gross domestic product per capita in Côte d’Ivoire) are regarded as highly cost effective. (Relative environmental contribution refers to the relative rate of excretion of eggs into the environment among preschool-aged children and school-aged children when compared with adults.)
Additional benefits
There is also a body of evidence supporting a causal link between schistosomiasis infection and incidence of both malaria and HIV. That is, by treating schistosomiasis through combination deworming, and thereby reducing immunodeficiency and female genital schistosomiasis, SCI may produce considerable additional benefits by reducing prevalence of both malaria and HIV.24252627 In particular, computational modelling by researchers at Yale University suggests that deworming may be cost-effective for these effects alone: Depending on the efficacy of MDA, the analysis suggests that averting one HIV case through treating schistosomiasis costs as little as US$51.68–259.31.2829 If we assume that 20 DALYs are lost per infected adult,30 this translates to US$2.58-12.97 per DALY31 averted (our calculation). When combined with the $/DALY estimates above, this may result in SCI’s cost-effectiveness improving even further - to somewhere between $1.95 and $11.21 per DALY averted, which is incredibly low. However, the figures from these studies most likely do not accurately reflect SCI’s impact in practice, and are not supported by a sufficiently large body of evidence for us to be confident of their accuracy (the relationship between deworming and HIV incidence is supported by a recent Cochrane review that suggests that deworming helminth co-infected individuals can delay HIV disease progression,32 but these estimates of cost-effectiveness are not). Still, they do suggest that SCI may be even more cost-effective than previously thought.
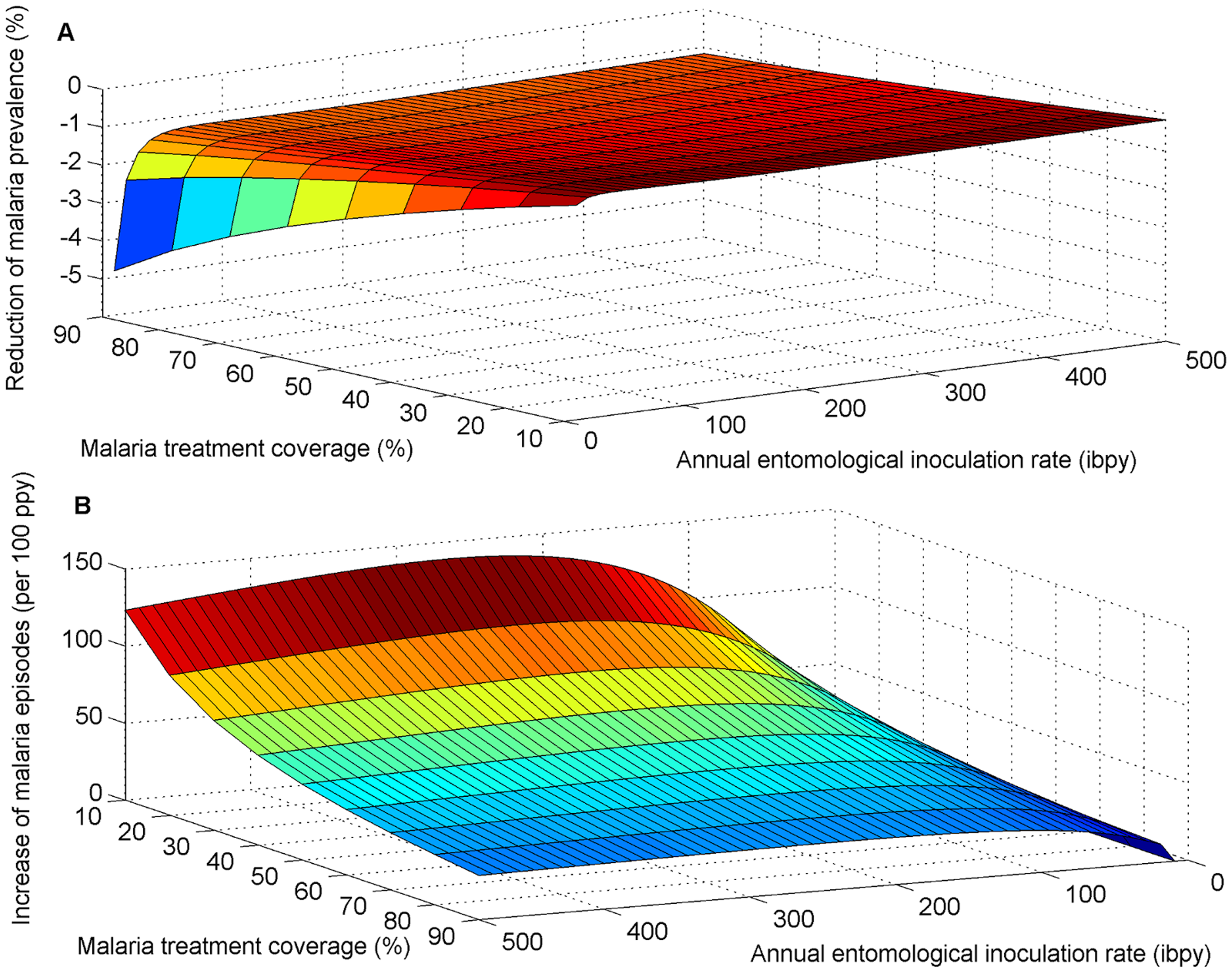
In addition, there is also some evidence that treating schistosomiasis also had the potential to reduce malaria prevalence.33 Depending on the context, MDA may reduce malaria prevalence by as much as 5% (see Figure 2 below).34 This relationship is supported by several other studies,35363738 and is believed to be causal due to the biological reasons for coinfection.394041 As the prevalence of both diseases (like that of HIV) is high in sub-Saharan Africa and a large proportion of the population is co-infected in the countries in which SCI operates, this again suggests that SCI may be considerably more cost-effective than has already been estimated above.
Figure 4: The impact of the interaction between S. mansoni and malaria on the effectiveness of MDA for reducing malaria prevalence and symptomatic malaria episodes.42
Possible negative impacts or offsetting
Would governments have funded the same programs otherwise?
This seems unlikely, as SCI generally does not work on programs which had already existed prior to their involvement.43 GiveWell has spoken to Ethiopia’s NTDs Program Manager who has commented that the program there would likely not have gone forward without SCI’s involvement.44 However, it is difficult to ascertain an answer to this question.
Could local populations be hostile to MDA programs?
It is entirely possible that local populations may be hostile to MDA programs due to lack of information about the deworming campaigns. SCI has encountered at least two such incidents of public discontent.4546 Since these incidents, SCI has reported that it has worked to improve public education about its programs.47
Are parasites developing resistance to praziquantel?
Researchers continue to be worried about the development of drug resistance to praziquantel, as it is the only drug currently recommended by the World Health Organization for the treatment and control of human schistosomiasis.48 Some signs of resistance of schistosomes against praziquantel have been observed in the laboratory, but in the field only isolated cases of decreased effectiveness of praziquantel have been observed so far.49 The National Institutes of Health has recently awarded a $3.5 million grant over five years to understand the genetic changes in the schistosomiasis parasite that lead to drug resistance.50
Are reinfection rates increasing?
A recent report51 argues that climate change will make temperatures more suitable for schistosomiasis transmission over the next 20 years, not only in terms of spread to other currently non-endemic countries, but also in terms of increasing the intensities of the disease in countries currently affected. The authors suggest that this increase in infection and reinfection intensity may reduce the impact of control and elimination programmes such as those undertaken by SCI. On the other hand, with the resulting increase in need and thus scalability of SCI’s programs, this might also mean an increase in their effectiveness.
Is there a negative impact on the wider health system?
A study of an integrated NTD control program in Mali found both positive and negative impacts on the wider health system.52 Positive impacts included increased funding for medical professionals. Negative impacts included distraction of staff from core activities and fragmentation of the monitoring and evaluation system. These effects were heterogeneous between different districts. Districts with more robust health systems tended to have more positive spillover effects. Limitations of the study include: it was qualitative research specific to Mali so may not be generalisable to other settings; it was an integrated NTD program, which might use a higher proportion of healthcare professional time (mass drug administration is largely staffed by community volunteers); and it was one of the first integrated NTD programs (evidence suggests that vertical health interventions become better integrated with health systems over time).53 Finally, this analysis did not account for fewer NTD related complication burdening the healthcare system after MDA.
Is vector control more effective?
A recently published study argues that historically, schistosomiasis control programmes which have focussed on vector control (mainly by controlling the snail population, as snails are an intermediate host of worms) have produced greater reductions of the prevalence of schistosomiasis than programmes that just deliver mass drug administration.[^fn-1001] However, the authors do not make any claims about the cost-effectiveness of snail control relative to MDA. They simply show that the average snail control programme has reduced schistosomiasis prevalence more than the average MDA programme, but if snail control programmes are more expensive we could still prefer to fund MDA as it reduces schistosomiasis more cost-effectively.
Robustness of evidence
Evidence suggest that combination deworming provides a variety of benefits - related to schooling and earning, severe symptoms and mortality and general health effects (see our full cause report).
In regards to SCI itself, prevalence and intensity studies have been conducted following SCI-supported treated programs in multiple countries. In these studies, the infection rates were measured at the same schools each year and it was found, in each case, that both prevalence and intensity fell over time. In schools treated in Niger, overall prevalence of schistosoma haematobium fell from 75.4% to 38% after one year and, the prevalence of intense infections fell from 21.8% to 4.6%.5455 In SCI’s Burundi pilot, prevalence of schistosoma mansoni dropped from 12.7% to 1.7% after 4 years and prevalence of hookworm from 17.8% to 2.7%.56 In Malawi, prevalence of schistosoma haematobium dropped from 9% at baseline to 6% at one year and 4% after two years.5758 Although it is certainly possible that these decreases are due to other causes, we agree with GiveWell that the short timespan and rapid change indicates that they are attributable to SCI’s operations.59 However, the data has limitations, due to worries about the representativeness of the areas chose and low follow up rates.
Coverage studies were also conducted in numerous countries (Uganda in 2014, Malawi in 2012 and 2014, Côte d'Ivoire in 2014, Mozambique in 2015, Zanzibar in 2015, and Zambia in 2015) to establish what proportion of the population had received the combination deworming treatment. These studies were administered as in-person surveys of a sample of households. Although these studies were subject to several problems and limitations,60 they have indicated that SCI has been quite successful in achieving a high degree of coverage in some areas. Median coverage for schistosomiasis treatment was 77% in Malawi (2012), 69% in Malawi (2014), 82% in Côte d'Ivoire (2014), 47% in Uganda (2014), 81% in Mozambique (2015), 93% in Zambia (2015), and 80% in Zanzibar (2015).61 Median coverage for STH treatment was 59% in Malawi (2012), 44% in Malawi (2014), 82% in Côte d'Ivoire (2014), and 87% in Zanzibar (2015).62
However, it should be stressed that GiveWell has only had access to data from about half of the countries SCI works in, and has only seen results for one year, even if SCI worked in the country for longer. Partly, this is due to the fact that SCI is sometimes unable to share the data due to agreements with the institutions it works with.
Track record
SCI has a very strong track record of implementing successful mass drug administration programs, starting the first large scale schistosomiasis deworming programs in Ethiopia, Ivory Coast and Mozambique.63 SCI has received large, multi million dollar grants in the past from the Gates Foundation, USAID, and DFID. Moreover, SCI is affiliated with Imperial College London, and the members of their team are recognised as experts on neglected tropical diseases.
In most of the countries that SCI operates in, the deworming programs appear to go according to plan. However, there have been considerable problems with deworming programs in three countries. In Liberia, due to the Ebola epidemic, all deworming operations had been temporarily suspended. In Yemen, due to political tension, a staff member had to be temporarily evacuated in 2014, but has now resumed work. In Zambia, drug delivery seems to be a continuing problem: SCI reported in April 2014 that, due to delays during 2013, treatment scheduled across 6 provinces only took place between January and March 2014. The remaining 4 provinces were scheduled to receive MDA during July 2014. However, then in December 2014, SCI reported that only “piecemeal treatments” – about 60,000 – were successfully delivered. Overall, we feel that, as there were only 3 out of 16 countries in which operations had problems, and most were due to fairly unpredictable reasons, SCI’s operations seem to run very well.
GiveWell has expressed concerns about SCI financial documents, and noted that, before 2016, it was unable to determine SCI’s total funding and the way it allocated resources across programs. During 2016, SCI has been able to offer better financial reporting to Givewell. GiveWell has also since learnt of two major accounting errors. About $333,000 donated to SCI through GiveWell was misallocated by Imperial College, and it had mistakenly been allocated to a different department inside the university. Moreover, SCI underestimated its resources by $1.5 million, which led GiveWell to overestimate SCI room for more funding. While GiveWell expressed its concern about these mistakes, it also stressed that it was a good sign that they were able to learn about them, and that communication with SCI has significantly improved in 2016.64
Room for more funding
SCI employs the funds collected in a given year to fund the following budget year. In April 2016, SCI has employed most of its unrestricted funding to the 2016/2017 budget year. GiveWell believes that any funds donated to SCI from now until February/March 2017 would be used for the organization’s 2017-18 budget, and that SCI would be able to use the funds productively. In addition, if SCI’s immediate funding gap was filled, it may be able to productively use additional funding to expand its operations into new countries and potentially use existing infrastructure to do mass drug administration against other diseases such as malaria. SCI communicated to GiveWell in October 2015 that their room for more funding for April 2017-March 2018 would be around $11.6 million. However, this is likely not to fully take into account funding that SCI is likely to receive from DFID, and the gap might thus be lower. Moreover, a series of factors (including changes in political circumstances, changes in the availability of drug supplies and coordination with other organizations) might significantly change the estimate.
One factor that affects SCI’s room for more funding in the long run is the global funding gap for MDAs as, with the number of people who are targeted to receive deworming rising (see Figure 5),65 SCI could most likely scale up and strengthen its efforts in the countries they currently operate in and expand to new countries. A recent report by the World Health Organisation estimates that the overall costs for all kinds of preventive chemotherapy (which means mass deworming and has no resemblance to chemotherapy used as cancer treatment) in the coming year are in the hundreds of millions (see 6).66 The investment target for preventive chemotherapy against schistosomiasis (for delivery only- medicines are donated by the pharmaceutical companies) during 2015–2020 is estimated to be about $59 million ($51–67 million) per year (see Figure 5). This already takes into account that countries such as Brazil and India completely covered the cost of distributing the donated medicines using their own resources. In poorer countries such as the DRC, Sengal and Niger,67 costs of distributions are shared among the local Ministries of Health and SCI, as well as other organisations such as the USAID, END Fund, CNTD, and APOC. But poor countries are not in a position to execute these programs by themselves and bigger aid givers such USAID are also do not completely fund such programs. Thus, it is very much conceivable that SCI, with their unique expertise, might play a substantial part implementing such future deworming programs and thus require substantially more funds. This could become subject to change in case bigger agencies such as USAID recognize the cost effectiveness of interventions targeting NTDs.
Figure 5: Number of people targeted for coverage with integrated preventive chemotherapy, selected NTDs (from recent WHO report on NTDs).68 Note that the dots indicate the number of people treated in 2012 and that the solid lines are targets not forecasts. Targets assume integrated delivery of preventive chemotherapy for LF and onchocerciasis in Africa, schistosomiasis and STH among school-age children, and LF and STH outside Africa. Pending further evidence, they do not yet assume further integration of LF and onchocerciasis in Africa with schistosomiasis and STH.
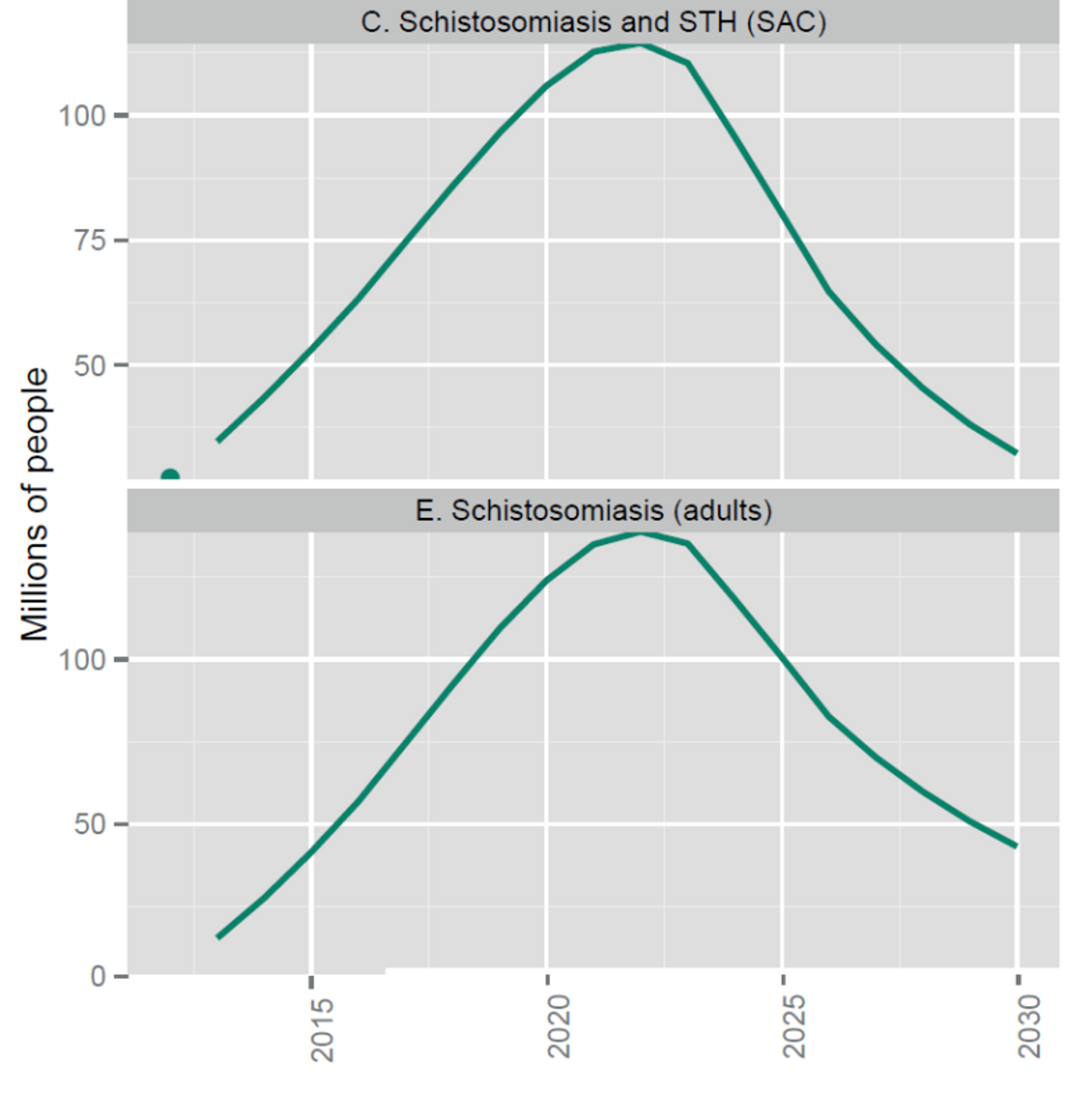
Figure 6: The overall projected costs of all preventive chemotherapy (i.e. mass drug administration against NTDs including schistosomiasis and soil transmitted helminths) in the coming years. This excludes medicine prices, which are donated by the pharmaceutical companies. Figure adapted from (Fig. 2.4 Investment targets for universal coverage against NTDs from recent WHO report on NTDs).69
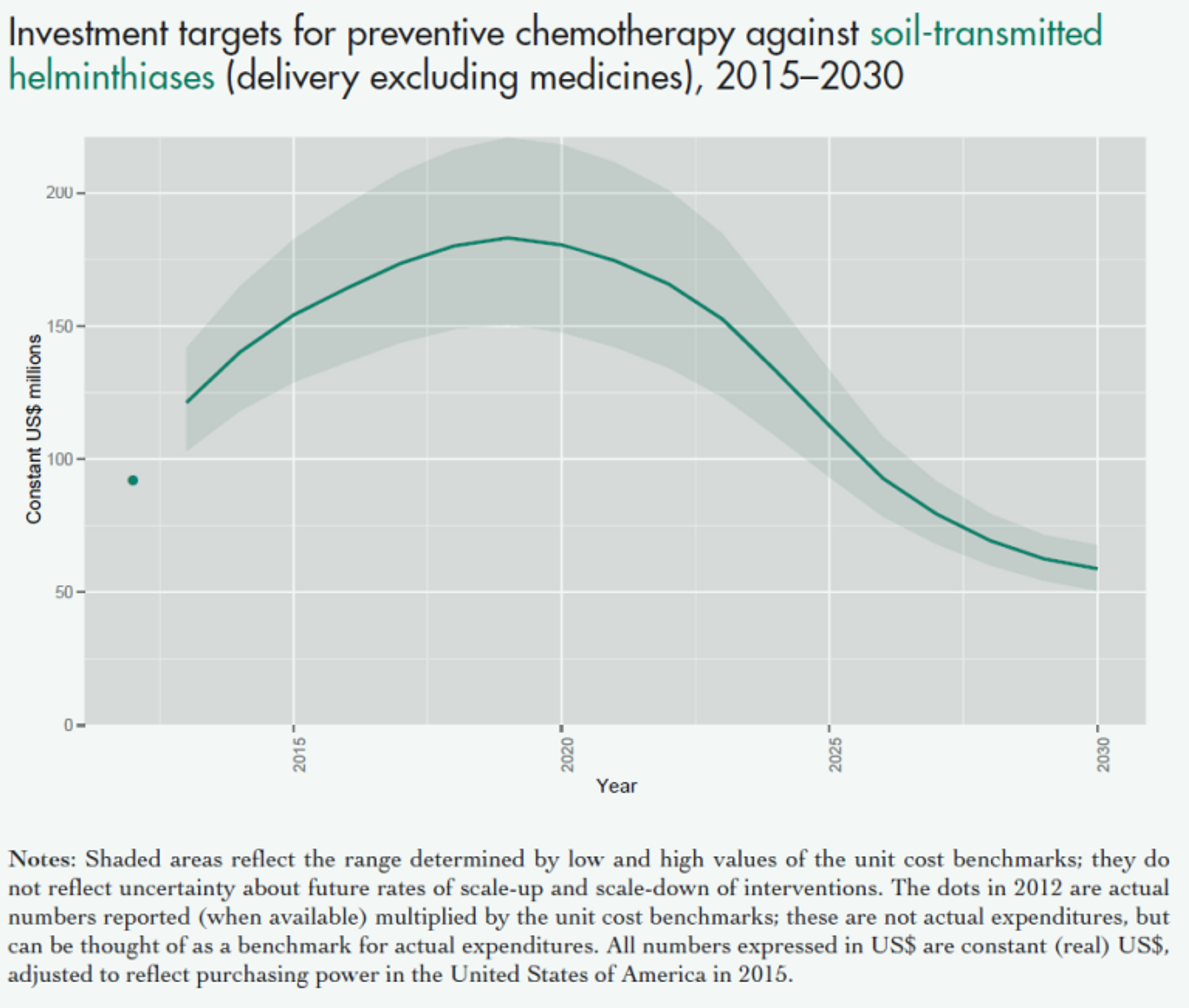
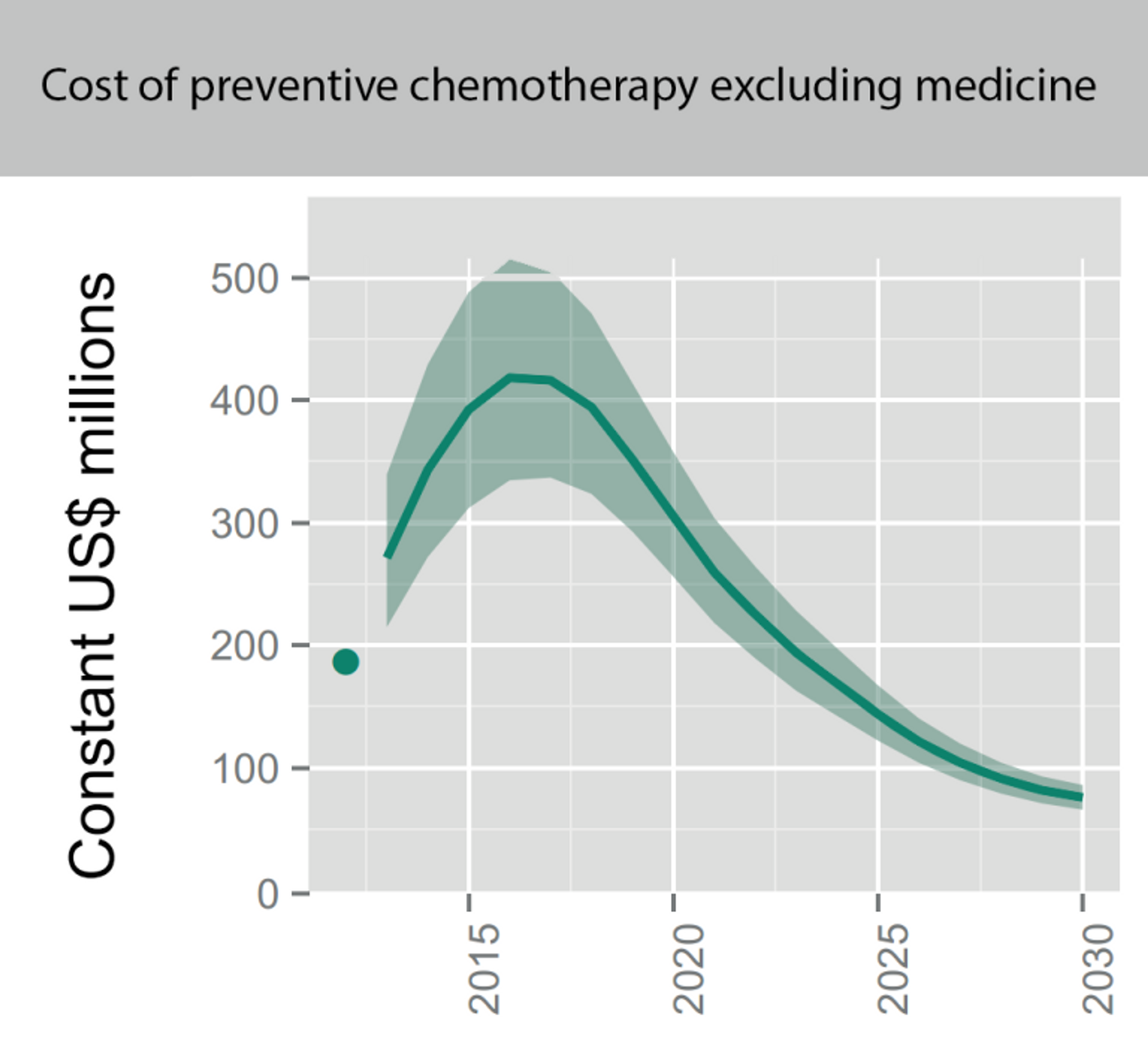
Figure 7: The projected costs of preventive chemotherapy against schistosomiasis (upper panel) and soil transmitted helminths (lower panel) in the coming years. This excludes medicine prices, which are donated by the pharmaceutical companies. Figure adapted from a recent WHO report on NTDs.70 Note that shaded areas reflect the range determined by low and high values of the unit cost benchmarks; they do not reflect uncertainty about future rates of scale-up and scale-down of interventions. The dots in 2012 are actual numbers reported (when available) multiplied by the unit cost benchmarks, which is the approximate cost of delivering one treatment; these are not actual expenditures, but can be thought of as a benchmark for actual expenditures. All numbers expressed in US$ are constant (real) US$, adjusted to reflect purchasing power in the United States of America in 2015.
Fundraising activities
SCI has been very successful at starting partnerships and receiving funding from governmental and non-governmental organisations. However, in recent years, one might think that they should have received more funding from private foundations and private donors, as one might expect based on from their effectiveness, reputation and room for additional funding. Based on our communications with SCI, they are currently working closely with the Imperial College Development Office team on increasing their efforts to attract grants from private foundations and philanthropists. They are also in the process of hiring additional staff for their Fundraising and Advocacy team. At Giving What We Can we assess charities based on their effectiveness and not exclusively based on overhead costs, so this use of resources to attract more funding does not significantly detract from our confidence in SCI’s cost-effectiveness.[^fn-1001]: "Global Assessment of Schistosomiasis Control Over the Past Century ..." 2016. 25 Jul. 2016 http://dx.plos.org/10.1371/journal.pntd.0004794