Tuberculosis
Giving What We Can no longer conducts our own research into charities and cause areas. Instead, we're relying on the work of organisations including J-PAL, GiveWell, and the Open Philanthropy Project, which are in a better position to provide more comprehensive research coverage.
These research reports represent our thinking as of late 2016, and much of the information will be relevant for making decisions about how to donate as effectively as possible. However we are not updating them and the information may therefore be out of date.
About a third of the Earth's population has tuberculosis, and a significant percentage of these people become sick or infectious during their lifetime. The disease can be combatted with immunisations and antibiotics; the latter in particular can be highly cost-effective. However, there is conflict between the DCP2's and the World Health Organisation's data for these interventions, and additional research is required.
Tuberculosis, or TB, is an infectious bacterial disease that most commonly affects the lungs. Like the common cold, it is transmitted from person to person via droplets from the throat and lungs of people with the active respiratory disease.
In healthy people, infection with tuberculosis often presents with no symptoms, since the person's immune system acts to 'wall off' the bacteria. The symptoms of active TB of the lung are coughing (sometimes with sputum or blood), chest pains, weakness, weight loss and fever. [1]
One third of the world's population is currently infected with TB. 5–10% of people who are infected with TB alone (i.e. are not co-infected with HIV) become sick or infectious at some time during their life. People with HIV and TB infection are much more likely to develop TB. [2]
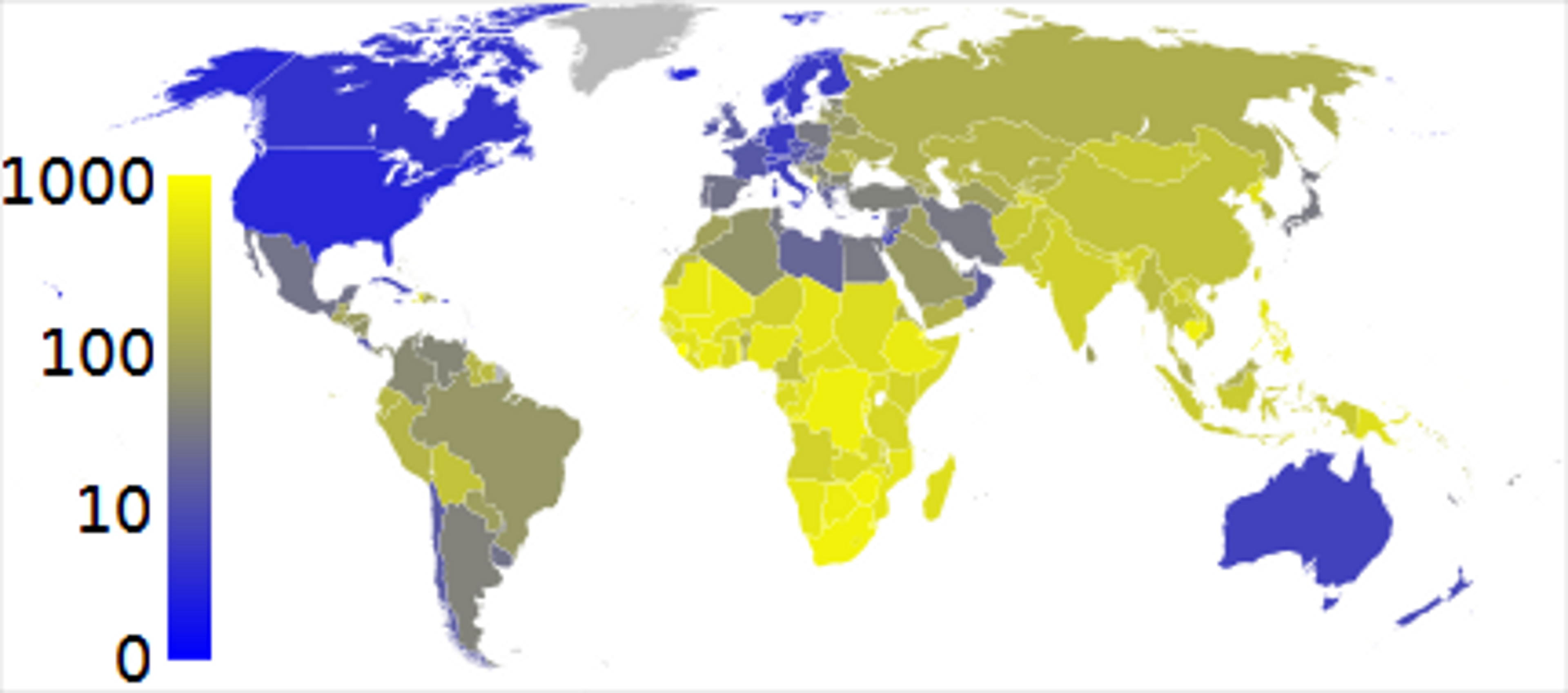
TB tends to affect the poorest countries, and is prevalent throughout Asia and Africa:
COST-EFFECTIVENESS
It is possible to immunise people against TB, using the BCG vaccine. Currently, however, the internationally recommended intervention is the DOTS (Directly Observed Treatment, Short course) strategy. This is a six-month supervised course of antibiotics.
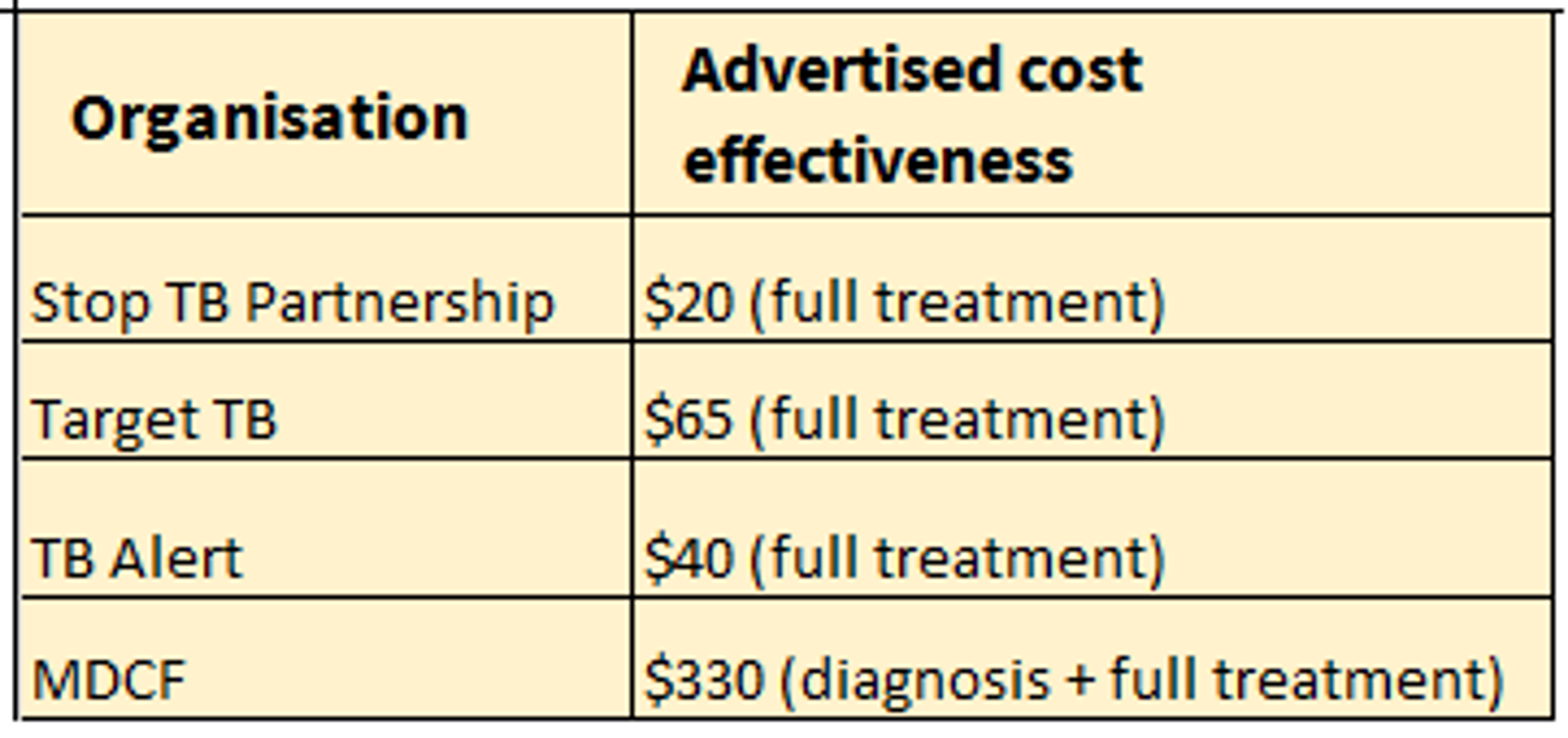
Cost-effectiveness estimates from charities that focus on TB are as follows:
The DCP2 and WHO-CHOICE cost-effectiveness estimates are as follows: [3]
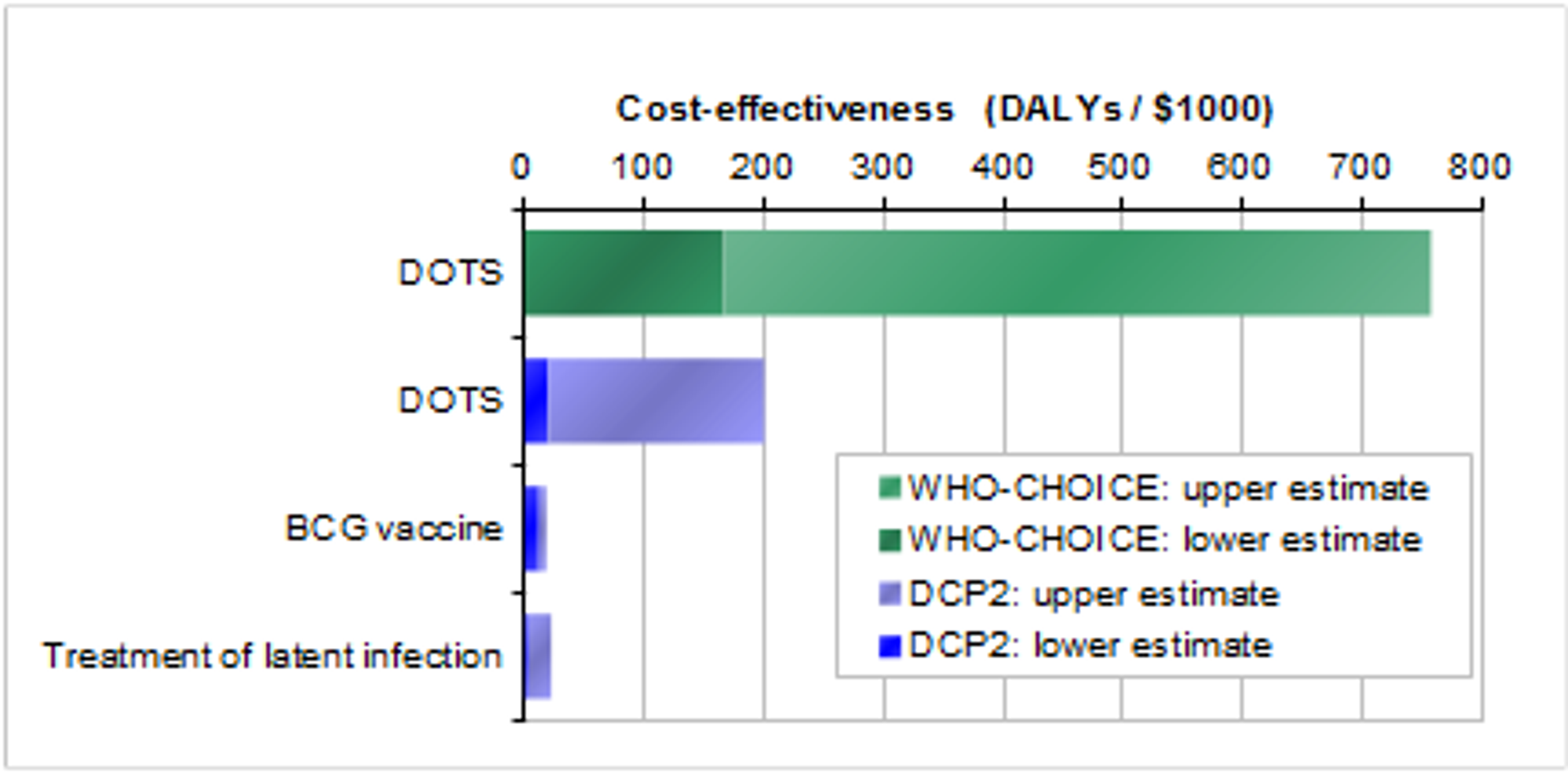
According to the DCP2 estimate, $1,000 spent on the DOTS program averts 125 Disability Adjusted Life Years, which is on a par with saving four lives. According to the WHO-CHOICE estimate, $1,000 spent on the DOTS program averts 750 DALYs, which is on a par with saving 25 lives.
We're unsure why there is such a large discrepancy between the figures from DCP2 and WHO-CHOICE. WHO-CHOICE's cost-effectiveness estimates for the DOTS strategy in other regions of the world also seem surprisingly high: this suggests that the WHO-CHOICE estimate may be optimistic. Overall, we're uncertain about exactly how cost-effective DOTS is (which makes it difficult to compare with other interventions), but there does seem to be agreement in the literature that the DOTS strategy is a highly cost effective intervention. [4]
OTHER INFORMATION
Are there side-effects?
Positive side-effects of DOTS include:
- Economic benefits: care is no longer needed for the patient, who is able to become economically productive again.
- Curing tuberculosis drastically reduces transmission rates: one person with active TB infects 15 people every year.
Possible negative side-effects include:
- The cost to the recipient of travel and working hours lost in order to receive the chemotherapy.
CONCLUSION
Charities that focus on the DOTS strategy appear very impressive. If the WHO-CHOICE estimates are correct, they are at the very top end of cost-effectiveness - about 100 times as effective as typical developing-world health interventions. There are some countervailing concerns, however: the DCP2 estimate of cost-effectiveness contradicts WHO-CHOICE's estimate, and the positive side-effects of treatment may therefore be lower than for the treatment of the seven most prevalent neglected tropical diseases.
For these reasons, we would recommend that donors seriously consider charities working in this area, but please bear in mind the uncertainty that surrounds this recommendation.
Of the many charities focussing on TB, we recommend the Stop TB Partnership.
Sources:
- WHO summary of tuberculosis.
- WHO tuberculosis factsheet.
- Note that the DCP2 estimates for DOTS comes from Box 16.1 on p. 301, not from the main cost-effectiveness table. The figure from Box 16.1 excludes Eastern and Central Europe, which we think gives a more accurate picture.
- DCP2 report, chapter 16, p. 296.
R. Baltussen, K. Floyd, C. Dye, 'Cost effectiveness analysis of strategies for tuberculosis control in developing countries', BMJ, doi:10.1136/bmj.38645.660093.68
Last updated: in or before 2012